Effect of pH on the bioprotection exerted by some strains of arbuscular mycorrhizal fungi
Main Article Content
Abstract
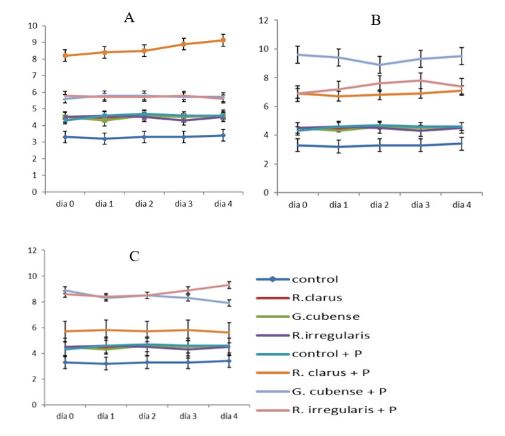
Arbuscular mycorrhizae have been widely described as favoring plant growth and making physical, biochemical and physiological changes in roots that lead to a better general condition of the plant and contribute to alleviate abiotic and biotic stress situations. As a result of their multiple benefits, their use in Cuban agriculture has been gradually increasing. INCA's generalist strain recommendation system is based, fundamentally, on the soil type and its associated fertility. Knowing how the bioprotection effect of different strains is integrated with pH can contribute to elucidate whether the effect is associated with a strain "per se" or depends on the effectiveness of the strains. For this purpose, an experiment was designed using a red-yellowish Argissolo soil, adjusting the Ca2+ and Mg2+ concentrations to a single level and three pH values (5.5; 6.5 and 7.2) with the aim of finding out whether differences in pH also influence the bioprotection exerted by these strains. Rhizophagus irregularis, Glomus cubense and Rizophagus clarus strains recommended for different pH ranges were used and Fusarium oxysporum f. sp. phaseoli was inoculated on 21-day-old bean plants as pathogen. Strains originated differentiated pH-dependent responses in the intensity of colonization, bioprotection exerted and active induction of peroxidases, indicating that the bioprotection effect was associated with the effectiveness of each strain at one or another pH.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Those authors who have publications with this journal accept the following terms of the License Attribution-NonCommercial 4.0 International (CC BY-NC 4.0):
You are free to:
- Share — copy and redistribute the material in any medium or format
- Adapt — remix, transform, and build upon the material
The licensor cannot revoke these freedoms as long as you follow the license terms.
Under the following terms:
- Attribution — You must give appropriate credit, provide a link to the license, and indicate if changes were made. You may do so in any reasonable manner, but not in any way that suggests the licensor endorses you or your use.
- NonCommercial — You may not use the material for commercial purposes.
- No additional restrictions — You may not apply legal terms or technological measures that legally restrict others from doing anything the license permits.
The journal is not responsible for the opinions and concepts expressed in the works, they are the sole responsibility of the authors. The Editor, with the assistance of the Editorial Committee, reserves the right to suggest or request advisable or necessary modifications. They are accepted to publish original scientific papers, research results of interest that have not been published or sent to another journal for the same purpose.
The mention of trademarks of equipment, instruments or specific materials is for identification purposes, and there is no promotional commitment in relation to them, neither by the authors nor by the publisher.
References
Rivero J, Gamir J, Aroca R, Pozo MJ, Flors V. Metabolic transition in mycorrhizal tomato roots. Frontiers in Microbiology [Internet]. 2015 [cited 05/07/2022]; doi:10.3389/fmicb.2015.00598
Lanfranco L, Bonfante P, Genre A. The Mutualistic Interaction between Plants and Arbuscular Mycorrhizal Fungi. Microbiology Spectrum. 2016;1-20. doi:10.1128/microbiolspec.FUNK-0012-2016
Pozo MJ, López-Ráez JA, Azcón-Aguilar C, García-Garrido JM. Phytohormones as integrators of environmental signals in the regulation of mycorrhizal symbioses. New Phytologist. 2015;205(4):1431-6. doi:https://doi.org/10.1111/nph.13252
Helgason T, Fitter AH. Natural selection and the evolutionary ecology of the arbuscular mycorrhizal fungi (Phylum Glomeromycota). Journal of Experimental Botany. 2009;60(9):2465-80. doi:10.1093/jxb/erp144
Smith FA, Smith SE. How harmonious are arbuscular mycorrhizal symbioses? Inconsistent concepts reflect different mindsets as well as results. The New Phytologist. 2015;205(4):1381-4.
Espinosa R, Felix F, Martinez L, Cañizares P, Yakelín R, Ortega E. Manejo, integración y beneficios del biofertilizante micorrízico EcoMic (r) en la producción agrícola [Internet]. 2020. INCA. Mayabeque, Cuba; 2020. 155 p.
Hashem A, Abd_Allah EF, Alqarawi AA, Egamberdieva D. Arbuscular Mycorrhizal Fungi and Plant Stress Tolerance. In: Egamberdieva D, Ahmad P, editors. Plant Microbiome: Stress Response [Internet]. Singapore: Springer; 2018 [cited 11/07/2022]. p. 81-103. doi:10.1007/978-981-10-5514-0_4
Lanfranco L, Fiorilli V, Gutjahr C. Partner communication and role of nutrients in the arbuscular mycorrhizal symbiosis. New Phytologist. 2018;220(4):1031-46. doi:https://doi.org/10.1111/nph.15230
Venturi V, Keel C. Signaling in the Rhizosphere. Trends in Plant Science. 2016;21(3):187-98. doi:10.1016/j.tplants.2016.01.005
Herrera-Peraza RA, Hamel C, Fernández F, Ferrer RL, Furrazola E. Soil-strain compatibility: the key to effective use of arbuscular mycorrhizal inoculants? | SpringerLink. 2011;21:183-193.
Wendland-Ferreira A. Comunicación personal EMBRAPA [Internet]. 2020 [19/07/2022]. Available from: https://www.embrapa.br/agencia-de-informacao-tecnologica/inicial
Fernández F, Dell’Amico J, Pérez Y. Inoculante micorrizógeno líquido. Oficina Cubana de la Propiedad Industrial. 2009;23479.
Pozo MJ, Cordier C, Dumas‐Gaudot E, Gianinazzi S, Barea JM, Azcón‐Aguilar C. Localized versus systemic effect of arbuscular mycorrhizal fungi on defence responses to Phytophthora infection in tomato plants. Journal of Experimental Botany. 2002;53(368):525-34. doi:10.1093/jexbot/53.368.525
Vierheilig H, Schweiger P, Brundrett M. An overview of methods for the detection and observation of arbuscular mycorrhizal fungi in roots†. Physiologia Plantarum. 2005;125(4):393-404. doi:https://doi.org/10.1111/j.1399-3054.2005.00564.x
Giovannetti M, Mosse B. An Evaluation of Techniques for Measuring Vesicular Arbuscular Mycorrhizal Infection in Roots. The New Phytologist. 1980;84(3):489-500.
Trouvelot A, Kough JL, Gianinazzi-Pearson V. Mesure du taux de mycorhization VA d’un système radiculaire. Recherche de méthode d’estimation ayant une signification fonctionnelle. In 1986 [cited 11/07/2022]. p. 217-21. Available from: http://pascal-francis.inist.fr/vibad/index.php?action=getRecordDetail&idt=8758731
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry. 1976;72(1):248-54. doi:10.1016/0003-2697(76)90527-3
Heitefuß R, Williams PH. Oxidative enzymes. In: HEITEFUSS, R., and P. H. WILLIAMS. In: Physiological Plant Pathology [Internet]. New York: Springer Science & Business Media; 2012. p. 617-27. Available from: https://books.google.es/books?hl=es&lr=&id=hfLuCAAAQBAJ&oi=fnd&pg=PA1&dq=Oxidative+enzymes.+In:+HEITEFUSS,+R.,+and+P.+H.+WILLIAMS+(eds.).+Physiological+Plant+Pathology.+Encyclopedia+of+Plant+Pathology&ots=fu0GAetnMC&sig=pBM1iYFG95tlKKzYoQXrcb4q7UQ#v=onepage&q&f=false
Sun-Xue G, Tang M. Comparison of four routinely used methods for assessing root colonization by arbuscular mycorrhizal fungi. Botany. 2012;90:1073-83. doi:10.1139/b2012-084
Rivera R, González PJ, Hernández A, Martín G, Ruiz L, Fernández K, et al. La importancia del ambiente edáfico y del pH sobre la efectividad y la recomendación de cepas eficientes de HMA para la inoculación de los cultivos. In: VIII Congreso de la Sociedad Cubana de la Ciencia del Suelo. 2015.
Kanwal S, Bano A, Malik RN. Role of arbuscular mycorrhizal fungi in phytoremediation of heavy metals and effects on growth and biochemical activities of wheat (Triticum aestivum L.) plants in Zn contaminated soils. African Journal of Biotechnology. 2016;15(20):872-83. doi:10.4314/ajb.v15i20
Chaudhary VB, O’Dell TE, Rillig MC, Johnson NC. Multiscale patterns of arbuscular mycorrhizal fungal abundance and diversity in semiarid shrublands. Fungal Ecology. 2014;12:32-43. doi:10.1016/j.funeco.2014.06.003
Alguacil M del M, Torres MP, Montesinos-Navarro A, Roldán A. Soil Characteristics Driving Arbuscular Mycorrhizal Fungal Communities in Semiarid Mediterranean Soils. Applied and Environmental Microbiology. 2016;82(11):3348-56. doi:10.1128/AEM.03982-15
Wild A. Condiciones del suelo y desarrollo de las plantas según Russell. In: La población microbiana del suelo. Mur di-Prensa. Madrid, España: Ed. Mundi-Prensa; 1992. p. 471-94.
Igiehon NO, Babalola OO. Below-ground-above-ground Plant-microbial Interactions: Focusing on Soybean, Rhizobacteria and Mycorrhizal Fungi. The Open Microbiology Journal. 2018;12:261-79. doi:10.2174/1874285801812010261
Jamiołkowska A, Księżniak A, Gałązka A, Hetman B, Kopacki M, Skwaryło-Bednarz B. Impact of abiotic factors on development of the community of arbuscular mycorrhizal fungi in the soil: a review. International Agrophysics. 2018;32(1):133-40. doi:10.1515/intag-2016-0090
Bücking H, Kafle A. Role of Arbuscular Mycorrhizal Fungi in the Nitrogen Uptake of Plants: Current Knowledge and Research Gaps. Agronomy. 2015;5(4):587-612. doi:10.3390/agronomy5040587
Choudhary KK, Chaudhary N, Agrawal S, Agrawal M. Reactive Oxygen Species in Plants: Boon Or Bane - Revisiting the Role of ROS. In: Reactive oxygen species: generation, damage and quenching in plants during stress. In: Singh V.P. Wiley, Hoboken. NJ, USA: John Wiley & Sons; 2017.
Ouzounidou G, Skiada V, Papadopoulou KK, Stamatis N, Kavvadias V, Eleftheriadis E, et al. Effects of soil pH and arbuscular mycorrhiza (AM) inoculation on growth and chemical composition of chia (Salvia hispanica L.) leaves. Brazilian Journal of Botany. 2015;38(3):487-95. doi:10.1007/s40415-015-0166-6
Öpik M, Zobel M, Cantero JJ, Davison J, Facelli JM, Hiiesalu I, et al. Global sampling of plant roots expands the described molecular diversity of arbuscular mycorrhizal fungi. Mycorrhiza. 2013;23(5):411-30. doi:10.1007/s00572-013-0482-2
Gilbert L, Johnson D. Chapter Four - Plant-Plant Communication Through Common Mycorrhizal Networks. In: Becard G, editor. Advances in Botanical Research [Internet]. Academic Press; 2017 [cited 19/07/2022]. p. 83-97. (How Plants Communicate with their Biotic Environment; vol. 82). doi:10.1016/bs.abr.2016.09.001
Lambers H, Albornoz F, Kotula L, Laliberté E, Ranathunge K, Teste FP, et al. How belowground interactions contribute to the coexistence of mycorrhizal and non-mycorrhizal species in severely phosphorus-impoverished hyperdiverse ecosystems. Plant and Soil. 2018;424(1):11-33. doi:10.1007/s11104-017-3427-2